 When an electron drops down between levels, it emits photons with the same amount of energy-the same wavelength-that it would need to absorb in order to move up between those same levels. Emission of Light by HydrogenĮlectrons can also lose energy and drop down to lower energy levels. Get the full hydrogen absorption infographic in Resource Gallery. This illustration shows a set of jumps that correspond to absorption of visible wavelengths (the Balmer Series). The shorter the wavelength, the higher the energy, and the higher the jump. An electron jumps from one energy level to another only when it absorbs a very specific wavelength of light (i.e., when it absorbs a photon with a specific energy). (Right) The relationship between the electron jumps and the specific wavelengths of light that the atom absorbs. 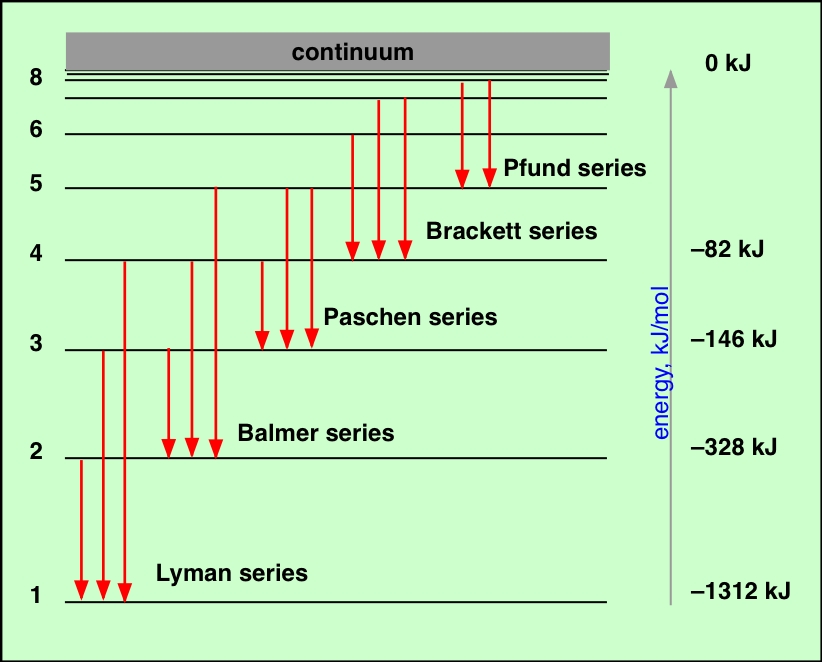
(Left) A simple model of a hydrogen atom showing four of the many possible “jumps” the electron could make when it absorbs light. The relationship between a hydrogen atom and its absorption spectrum. The shortest wavelength/highest energy light (violet 410 nm) causes the electron to jump up four levels, while the longest wavelength/lowest energy light (red 656 nm) causes a jump of only one level. Each of the absorption lines corresponds to a specific electron jump. In the visible part of the spectrum, hydrogen absorbs light with wavelengths of 410 nm (violet), 434 nm (blue), 486 nm (blue-green), and 656 nm (red). The absorption spectrum of hydrogen shows the results of this interaction. (Remember when we said that photons only carry very specific amounts of energy, and that their energy corresponds to their wavelength?) Said in another way, electrons absorb only the photons that give them exactly the right energy they need to jump levels. The energy that an electron needs in order to jump up to a certain level corresponds to the wavelength of light that it absorbs. In addition, it takes a very discrete amount of energy-no more, no less-to move the electron from one particular level to another. The interesting thing is that the electron can move only from one energy level to another. It can jump one level or a few levels depending on how much energy it absorbs. When the atom absorbs light, the electron jumps to a higher energy level (an “excited state”). When a hydrogen atom is just sitting around without much energy, its electron is at the lowest energy level. It consists of a single proton in the nucleus, and one electron orbiting the nucleus. Absorption of Light by HydrogenĪ hydrogen atom is very simple. 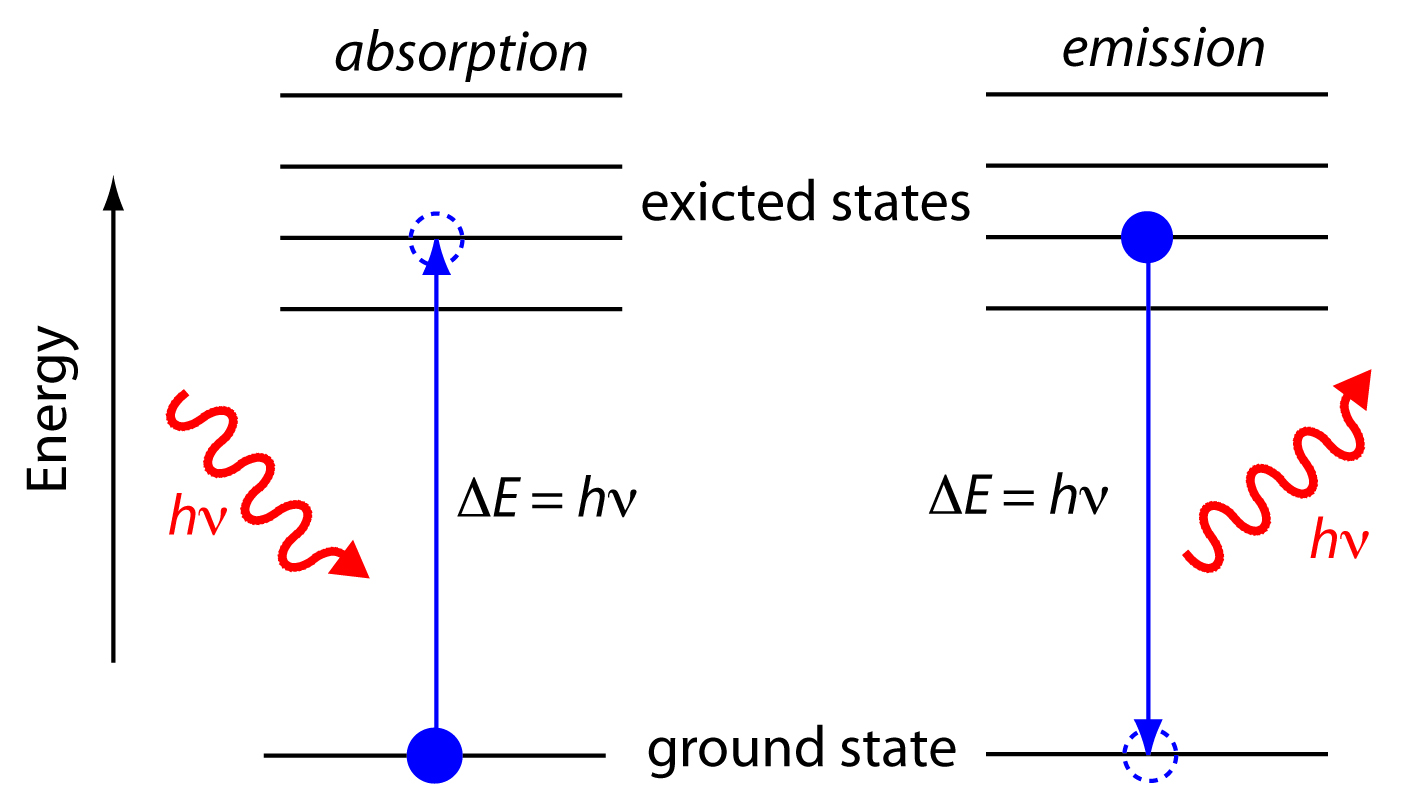
Why is this? Let’s take a look at hydrogen, the most abundant element in the universe. We can do both of these because each element has its own unique spectrum.Īn element’s spectrum is like its fingerprint, its autograph, its barcode. We can use a glowing nebula’s emission spectrum to figure out what gases it is made of based on the colors it emits. 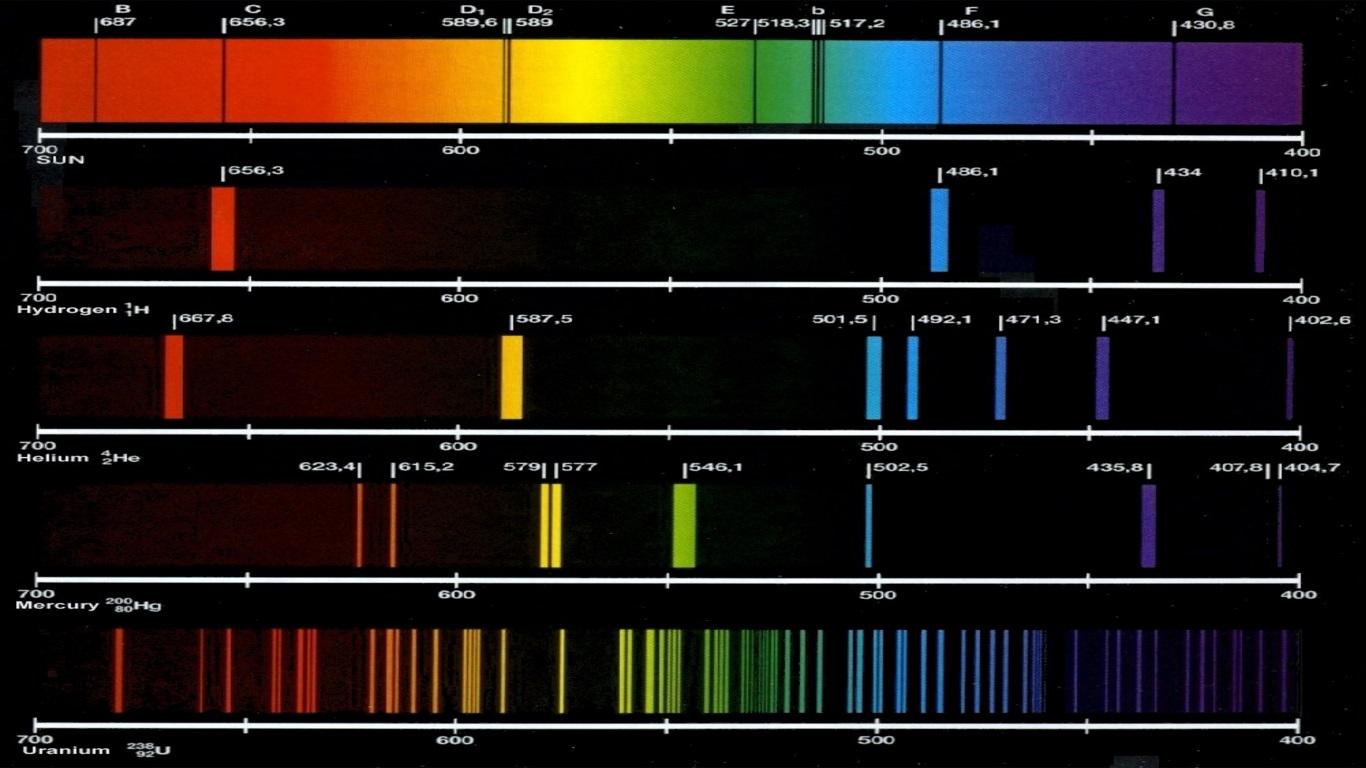
We can use a star’s absorption spectrum to figure out what elements it is made of based on the colors of light it absorbs. mostly all textbooks on the spectral lines of elements tend to only go up to uranium, however, textbooks go into much more detail for each element.Let’s go back to simple absorption and emission spectra. You can see a list of of all the known spectral lines of elements on Wikipedia since it seems to have the most updated table. These heavier elements likely have their own spectral lines, however, due to all I stated above, it isn't exactly possible to measure. This isn't even considering how much this would cost. This would make measuring their spectral lines nearly impossible. It's incredible because some of the heavier and more unstable elements have insanely short half lives ranging from 100.5 days (the most stable isotope of fermium (Fm, 100)), to 0.69 microseconds (0.00069 milliseconds) (Oganesson (Og, 118)). I could not find any data as to why this may be, however, I believe it is simply because we could not test for the spectral lines of the heavier elements due to their instability and scarcity. Also, tables of the spectral lines of elements only seem to go up to the 99th element, Einsteinium (not including Astatine (At, 85) and Francium (Fr, 87). However, it is much more difficult to observe the spectral lines of liquids and solids due to how close together the atoms are. 
Each element has its own distinct spectral line and this fact can and has been used to identify an element. Actually, this phenomena can be observed with liquids and solids as well. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
